Lüthje-Lab
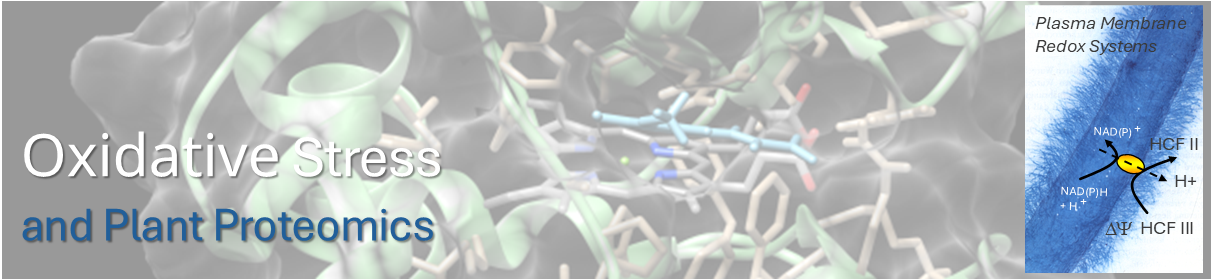
The plasma membrane builds the outer permeability barrier of the cell. It presents one of the most complex subproteomes that achieves important functions in cell signaling, transport processes, plant development and stress response. About 50 years ago, a constitutive transmembrane electron transport system - the so-called standard system - was demonstrated for plasma membranes of eucaryotic cells by the reduction of ferricyanide (HCF III).
Besides the standard system, a second, constitutive and iron inducible transmembrane electron transport system - the so-called turbo system or ferric-chelate reductase (FRO) - was demonstrated in root plasma membranes of non-grass monocots and dicots under iron-deficiency.
Several b-type cytochromes and NAD(P)H oxidoreductases could be identified in highly enriched plasma membrane fractions prepared by aqueous polymer two-phase partitioning (APTPP). The structure, electron transfer mechanism and function of plasma membrane-bound redox systems are one of our major topics.
References
S. Lüthje, B. Möller, F.C. Perrineau, K. Wöltje (2013) Redox pathways in plant plasma membranes and oxidative stress. Antioxidants and Redox Signaling 18:2163-83.
Lj. Menckhoff, N. Mielke-Mielke-Ehret, F. Buck, M. Vuletic, S. Lüthje (2013) Plasma membrane-associated malate dehydrogenase of maize (Zea mays L.) roots: Native versus recombinant protein. Journal of Proteomics 80C:66-77.
A. Bérczi, S. Lüthje, H. Asard (2001) b-Type cytochromes in plasma membranes of Phaseolus vulgaris hypocotyls, Arabidopsis thaliana leaves and Zea mays roots. Protoplasma 217:50–55
S. Lüthje, P. Van Gestelen, M.C. Córdoba-Pedregosa, J.A. González-Reyes, H. Asard, J.M. Villalba, M. Böttger (1998) Quinones in plant plasma membranes – a missing link? Protoplasma 205:43–51
S. Lüthje, O. Döring, S. Heuer, H. Lüthen, M. Böttger (1997) Oxidoreductases in plant plasma membranes (Review). Biochimica et Biophysica Acta 1331:81–102
An increasing evidence suggests association of plasma membrane redox components with microdomains (so-called lipid rafts) and protein-protein interactions. Beside physicochemical studies and lipid analysis of plasma membranes, proteomic approches were established in our laboratory to investigate the participation of redox proteins in putative protein assemblies. Both, blue native and clear native electrophoresis (hrCNE) confirmed an interaction of redox proteins with high molecular mass protein assemblies. Composition of those complexes depends on the state of development, physiological conditions and plant material investigated.
References
S. Lüthje, D. Hopff, A.K. Schmitt, C.-N. Meisrimler, Lj. Menckhoff (2009) Hunting for low abundant redox proteins in plant plasma membranes. Journal of Proteomics 72: 475-483
S. Lüthje (2008) Plasma membrane redox systems: Lipid rafts and protein assemblies. In: Progress in Botany 69 (eds. Lüttge et al.), pp. 169-200, ISBN-978-3-540-72953-2, Springer-Verlag, Berlin, Germany
P. Sperling, S. Franke, S. Lüthje, E. Heinz (2005) Glucocerebrosides are not the predominant sphingolipids in plant plasma membranes. Plant Physiology and Biochemistry 43:1031-1038
M. Bohn, E. Heinz, S. Lüthje (2001) Lipid composition and fluidity of plasma membranes isolated from corn (Zea mays L.) roots. Archives of Biochemistry and Biophysics 387:35–40
Further Topics
Class III peroxidases (secretory pathway)
Our team was the first that identified membrane bound heme peroxidases in plant plasma membranes. Class III peroxidases are a multigene family with at least 158 isoenzymes in maize. It appears that these enzymes build a cellular network of ROS scavenging and producing enzymes (PeroxiNET) that has to be tightly regulated. We established protocols for cell fractionation and gel-based proteomics to investigate the regulation and function of peroxidases within this network in plant development and oxidative stress. Beside studies on the structure and localization of these proteins by in silico analysis and GFP-fusions, heterologous expression and gene silencing (RNAi) are used for functional analysis.
S. Lüthje, T. Martinez-Cortes (2018) Membrane-bound class III peroxidases: unexpected enzymes with exciting functions. Internatl. J. Mol Sci 19, 2876
A. Mika, M.J. Boenisch, D. Hopff, S. Lüthje (2010) Membrane-bound guaiacol peroxidases are regulated by methyl jasmonate, salicylic acid, and pathogen elicitors. Journal of Experimental Botany 61: 831-841
A. Mika, F. Buck, S. Lüthje (2008) Membrane-bound class III peroxidases: Identification, biochemical properties and sequence analysis of isoenzymes purified from maize (Zea mays L.) roots. Journal of Proteomics 32:412-424
A. Mika, S. Lüthje (2003) Properties of guaiacol peroxidase activities isolated from corn (Zea mays L.) root plasma membranes. Plant Physiology 132:1489–1498
Heavy Metals
Studies of our team showed alterations in the plasma membrane proteome of iron uptake strategy I and strategy II plants (pea and maize) by both iron deficiency and iron toxicity for the first time. Peroxidases, redox systems and several other proteins are regulated by these stressors. The combination of iron deficiency with elicitor treatment support important functions of heme containing enzymes, but also of other plasma membrane proteins, in pathogen response.
D. Hopff, S. Wienkoop, S. Lüthje (2013) The plasma membrane proteome of maize roots grown under low and high iron conditions. Journal of Proteomics 91:605-18.
C.N. Meisrimler, S. Planchon, J. Renaut, K. Sergeant, S. Lüthje (2011) Alteration of plasma membrane-bound redox systems of iron deficient pea roots by chitosan. Journal of Proteomics, 74: 1437-1449
Emissions of industrial activities cause accumulation of cadmium and other toxic elements in the environment. Ions of these elements are taken up by unspecific transporters. Inside the cell, they cause oxidative stress and thereby changes in the cellular redox state. The molecular mechanisms of these reactions are not very well understood. So far, total peroxidase activity of samples is used as a general stress marker for many factors. My team established protocols to study the regulation of different peroxidase isoenzymes side by side in the same sample. We found that abundance and activity of different soluble, cell wall and membrane bound class III peroxidases changed under cadmium exposure.
S. Lüthje, A.G. Yilmaz, K. Ramanathan, W. Gräfenstein, J.M. Tabbert, S. Wienkoop, K. Heino, F.C. Perineau, S. Harder (2026) Cellular responses of maize roots to long-term cadmium exposure: Adjustments of class III peroxidases, plasma membrane and tonoplast sub-proteomes. Proteomes 14 (1), 11.
Weather extremes
Due to changing climate heat, drought and flooding events increases in northern hemisphere. Additionally, the use of heavy machinery in agriculture cause soil compaction, which supports waterlogging. Flooding provokes alterations in pH and oxygen levels (hypoxia and anoxia) in the soil and thereby iron toxicity. A recent study of our team showed alterations in abundance and activity of soluble class III peroxidases in maize by waterlogging.
A. Hofmann, S. Wienkoop, S. Lüthje (2022) Hypoxia-induced aquaporins and regulation of redox homeostasis by a trans-plasma membrane electron transport system in maize roots.Antioxidants 11 (5), 836.
A. Hofmann, S. Wienkoop, S. Harder, F. Bartlog, S. Lüthje (2020) Hypoxia-responsive class III peroxidases in maize roots: soluble and membrane-bound isoenzymes. Internatl. J. Mol. Sci. 21 (22), 8872
C.N. Meisrimler, F. Buck, S. Lüthje (2014) Alterations in soluble class III peroxidases of maize shoots by flooding stress. Proteomes 2 (3), 303-322